The cellulose of a tree's cell walls is then created using the glucose. Trees absorb carbon dioxide, and through the chemical process of photosynthesis, the carbon is removed from the CO 2 molecule and combined with a glucose molecule. In a carbon dioxide (CO 2) molecule, the carbon atom is initially joined to the oxygen atom. Consider an atom of carbon to demonstrate how an element is unchangeable. The nucleus of an atom cannot be changed without a nuclear reaction. The nucleus of an atom cannot be changed or changed by chemical reactions such as burning or bonding into atoms. The nucleus of an atom is held together by the strong nuclear force, which is challenging to overcome. The quantity of protons and neutrons in an element's nucleus determines its nature and physical characteristics. How are components made?Ĭhemical conversion of one element into another is not possible. As atoms link together to form molecules, they are the fundamental units of chemistry. The remaining 24 elements, which are referred to as man-made elements since they can't endure lengthy periods of stability, were created in particle accelerators. In order to create common table salt, sodium (Na) and chlorine (Cl) can establish an ionic bond in which the sodium loses an electron (becoming Na+) and transfers it to the chlorine atom (becoming Cl-).ĩ4 of the 118 distinct elements on the periodic table are known to occur in nature. As an illustration, sodium (Na) is a very reactive element. The amount of electrons in a given element can also change, leading to the formation of ions. /close-up-of-periodic-table-760294957-5a00794822fa3a0037921324.jpg)
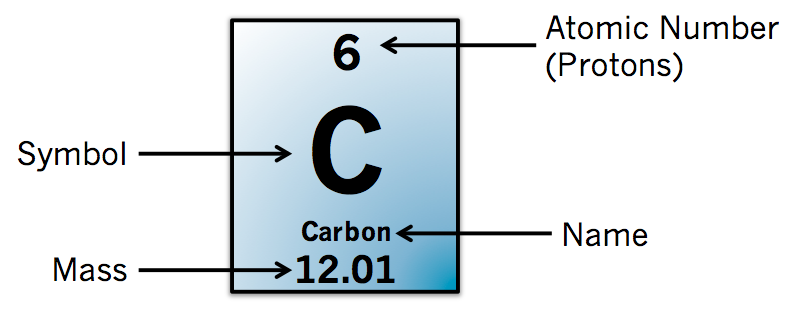
However, all three of them fall under the category of hydrogen. Deuterium contains one proton and one neutron, tritium has one proton and two neutrons, but normal hydrogen has one proton and no neutrons. Isotopes will have different physical properties although sharing the same chemical ones. Isotopes can be produced depending on the amount of neutrons in the nucleus. The atomic number, or quantity of protons in an element's nucleus, is used to categorise each element. Form molecules, atomic elements establish bonds. The Latin name of the element may serve as the basis for the symbol rather than the English name. Every element has a symbol and an atomic number. The chemical elements are arranged on the periodic table of the elements. An element's distinctive characteristics are determined by the number of protons in its nucleus. The foundation of chemistry and matter are elements. Mendeleev's reasoning for a periodic table based on the atomic weights of the elements was in disarray, and visualising the atom was a fight. Dmitri Mendeleev and other influential figures were seriously discussing elements lighter than hydrogen and those between hydrogen and helium. How elements came to be defined correctly?Ĭhemistry and physics were in disarray in 1913. Atomic weight is a reflection of the weight of the total number of neutrons and protons of the atom. Another key characteristic is atomic weight. /sequences-of-digits-172172089-59e629f9c41244001148b4a2.jpg)
For two atoms to be the same element, they must have the same number of protons, which will also mean they have the same atomic number. However, it's not enough to know that elements have electrons, protons, and neutrons. An element is a separate component of a bigger system or set in computing and mathematics. An element is a substance that cannot be broken down by non-nuclear reactions in chemistry and physics. A fundamental object that is difficult to divide into smaller bits is known as an element. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
